Definition: The word “Ion” originates from the Greek language and means “going” or “to go”. The term was first introduced to science by Michael Faraday (the famous physicist and chemist) in 1834 to define how electricity can move through a fluid (either a liquid or gas) rather than a conductive material like copper wire.
Unless you’re familiar with chemistry or physics, the subject of ionisation (ionization) can be quite complicated, so we’ve split this article into two levels:
Air Ions (very brief description).
The very simplest way to visualise an air ion, is to imagine it as being a tiny charge of “static” electricity carried within the air. This “static” can be either positive or negatively charged. (Hence positive or negative ion).
However, these charged “particles”, or ions, are not merely something floating in the air, they’re actually part of the ‘fabric’ of the air itself.
So as we breathe, they are drawn into our lungs and absorbed into the bloodstream. The most beneficial ions for us humans (and other animals too) are negative ions – in particular the negative ions of oxygen.
Air Ions – in more depth.
Air, like all matter, consists of individual atoms that bond together in different combinations to form molecules of the various gases that make up our atmosphere, (mainly: Nitrogen, Oxygen, Argon and Carbon dioxide)
In turn, each atom is made from even smaller particles called PROTONS (which have a positive charge) and ELECTRONS (having a negative charge).
Normally there are NEUTRONS too – which, as their name suggests, are neutral, having no electrical charge. (Since we are only concerned with electrical properties, we can ignore the neutrons at this time).
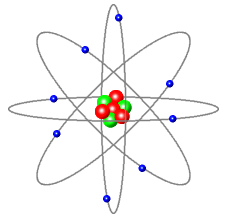
This diagram shows:
Blue: Electrons – with a negative charge.
Red: Protons – with a positive charge.
Green: Neutrons – with no charge.
So we have:
- Protons forming the centre (nucleus) of the atom.
- Electrons orbit this nucleus.
- Under normal circumstances there are equal numbers of each so their charges cancel out. The atom has no overall electrical charge.
However, the negatively charged electrons are very much lighter (in fact, almost two thousand times lighter) and under some circumstances can quite easily become dislodged. This upsets the electrical “balance” and leaves the atom with an overall positive charge. It becomes a “positive ion”.
Sometimes a “free” electron will attach itself to an atom which will tip the electrical balance the opposite way and make it a “negative ion”